Table of Contents
Pharma Aggregation is a new opportunity in Supply Chain drug traceability.
It has been considered as the next logical step after serialization by the industry expert, especially when the single package serialization is identified as proofed protection against counterfeiting and drug integrity.
The USA’s Drug Supply Chain Security Act (DSCSA) has put a significant change in the power of medicines from the beginning. It was created with the aim of removing up to 60%, counterfeit drugs, but DSCSA serves an additional purpose. It ensures that the gap-matches for each pharmaceutical product and establishes step-by-step instructions that pharma companies in the supply chain should follow.
As a pharmaceutical industry, $ 1 trillion worldwide manufacturers in India and China pack packaging in Southern Europe, products counterfeit is important for pharmaceutical suppliers anywhere in the United States, Asia, and around the world. For more important to protect integrity at non-essential levels as you have many opportunities to enter the sale of goods.
Aggregation Number: “The Packaging Identifier”
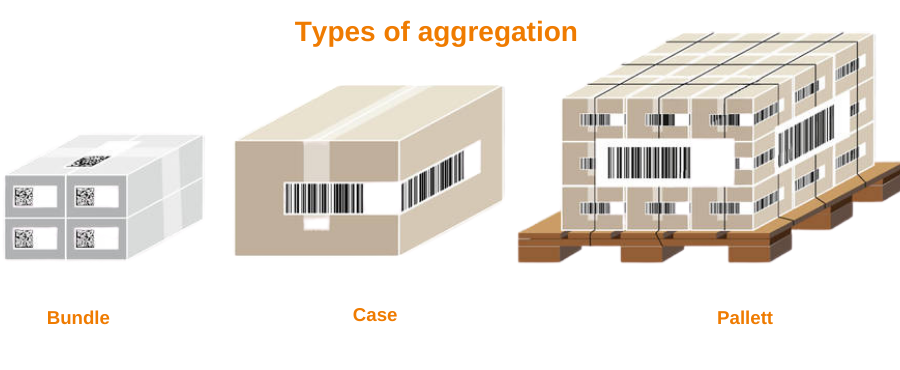
Moving one step from serialization, the process of connecting objects with different numbers to a higher-level layout, and also a unique serial number. They are called “parents”. For example, each hydroxychloroquine unit has a different identifier, and each lot has a label with a unique serial number. In this case, the container is childish and box-shaped.
How Does the Aggregation Work?
The pharmaceutical aggregation by its definition associates uniquely serialized medical items (medicines) to higher packaging levels with a unique serial number. The unit-serialized items come to a larger container (case, bundle, or pallet). In a bundle of bottles or cartons, each unit has its own serial number, but the bundle itself has a top label including a unique serial number. In short, pharma aggregation is the process of creating a hierarchical relationship between unique identifiers assigned to packaging containers.
Shared Benefits
Some of the largest pharmaceutical chains used are due to the significant benefits that are provided for integration in product track and trace. Some of these are:
Better Design Management: Considering the information collected by warehouse managers during the vial inspection and resolving bulk delivery delivered to the second store, helps you quickly find items (packs) while searching
Quick recycling: need to change the collection of certain bottles? It is very convenient due to an easy system’s drug locating. All you have to do is take the medicine out of it, get a different diagnosis, and change the location of the drug
Easy access to all individual “child” information with a “parent” batch scan
Fast drop shipping charges: Palette can be demolished for bulk shipping without the need for actual presence. All you must do is find the damaged parental property and keep it
Flexibility in Regional requirements: Aggregation facilitates the introduction of complex and chain-imported drugs. Many countries, including the United States, are expected to adopt pharmaceutical aggregation as part of their regulatory track and trace compliances.
Aggregation can save a lot of time because you usually don’t have to open collections or cases to scan individual “kids”. With access to all your data, it’s easy to track your products in every transaction, and your entire distribution system works efficiently, securely, and reliably.
Is Aggregation Mandatory?
Let’s face it! The US DSCSA and FMD (Counterfeit Drugs Directive) of the European Union do not collect or use aggregated data. However, this is recommended because it enhances procurement integrity and the above reasons and benefits.
Source: https://www.pharmiweb.com/article/why-is-pharma-aggregation-the-future-of-traceability-an-inside-story